PO43- Lewis Structure (Phosphate Ion) | PO43- Lewis Structure (Phosphate Ion) Did you know that Phosphorus can have expanded orbitals and can accommodate more than 8 electrons in its outer... | By
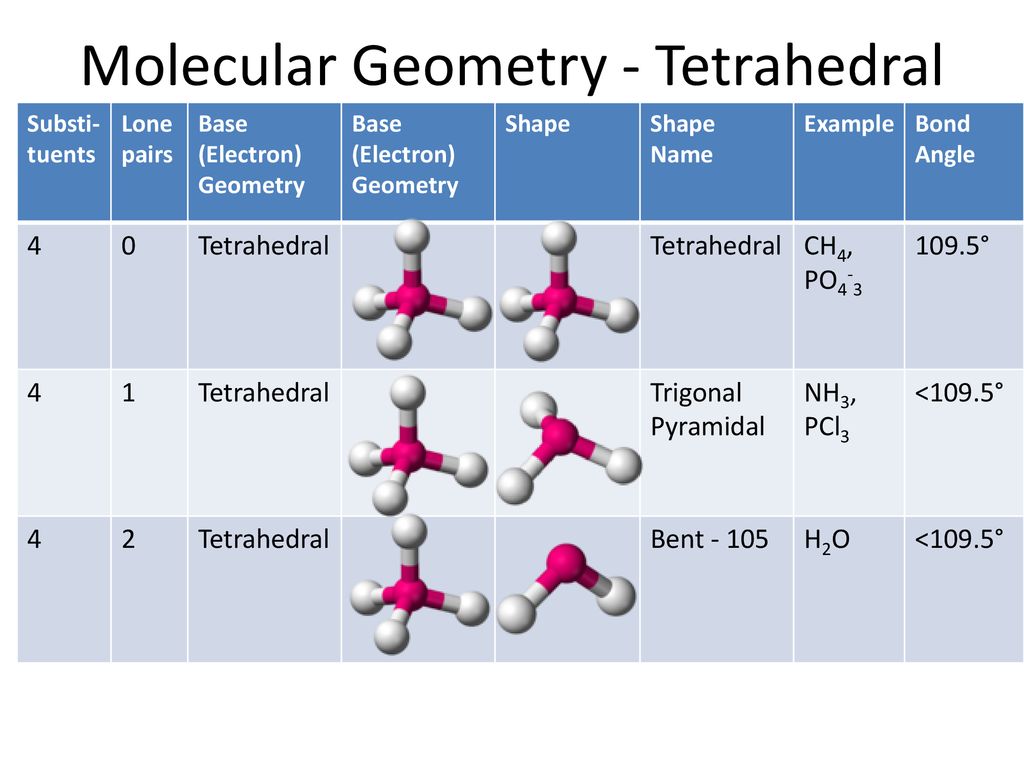
SOLVED: Molecular e) CS2 f) PI3 g) CF4 h) Br2CO (C is central) i) H2S j) PO43- k) SO32- 2) For compounds e) through i) above: a) Give the molecular geometry. b)

Is SF6 Polar or Nonpolar? - Polarity of Sulfur hexafluoride | Molecular geometry, Polar, Electron configuration
![SPOILER] [AAMC Official Guide Questions C/P Question #16] Is PO4(3-) polar or non-polar? EK says it is insoluble... AAMC says otherwise : r/Mcat SPOILER] [AAMC Official Guide Questions C/P Question #16] Is PO4(3-) polar or non-polar? EK says it is insoluble... AAMC says otherwise : r/Mcat](https://preview.redd.it/5whv97g4py811.png?width=640&crop=smart&auto=webp&s=5d77c21a9f0b544f925d587c7e01cf76511f1513)
SPOILER] [AAMC Official Guide Questions C/P Question #16] Is PO4(3-) polar or non-polar? EK says it is insoluble... AAMC says otherwise : r/Mcat



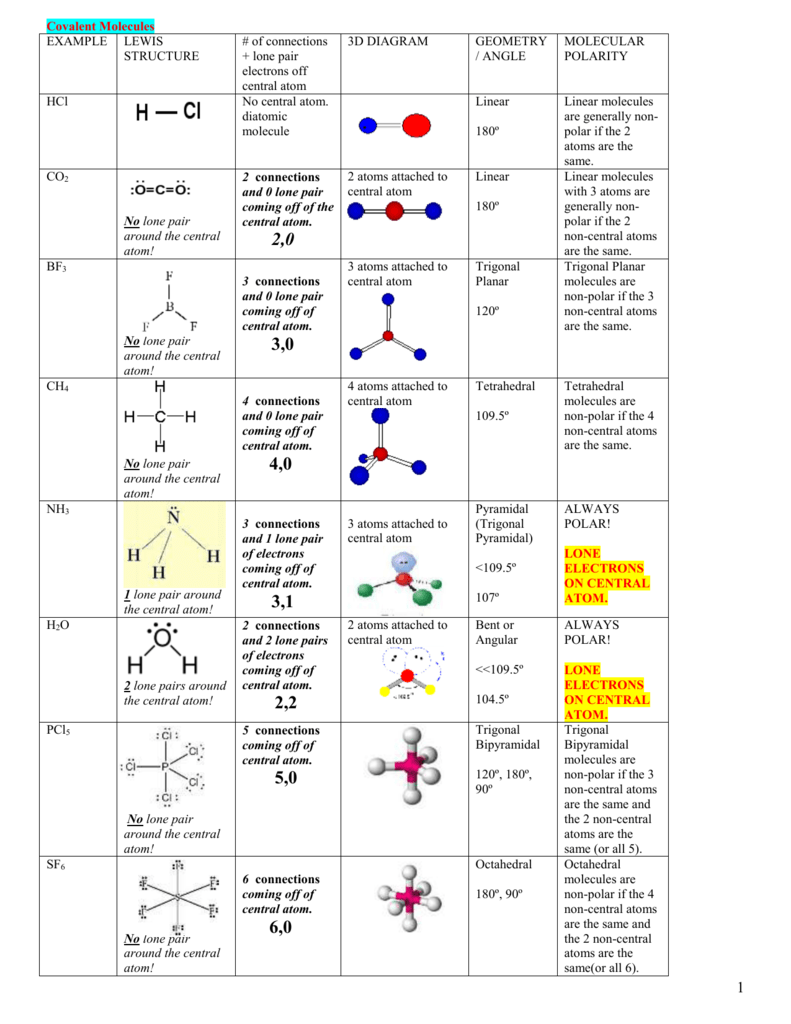


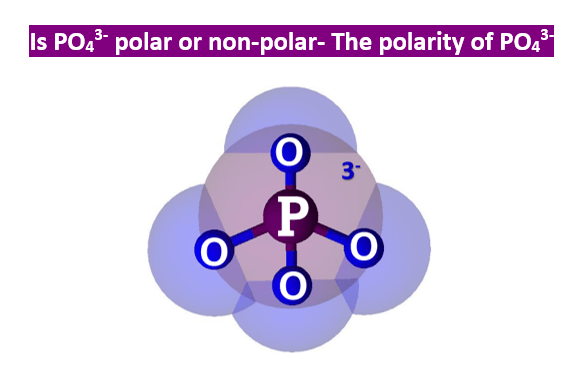
![PO4]3- - Phosphate PO4]3- - Phosphate](http://www.chemtube3d.com/images/gallery/inorganicsjpgs/po4_3-.jpg)