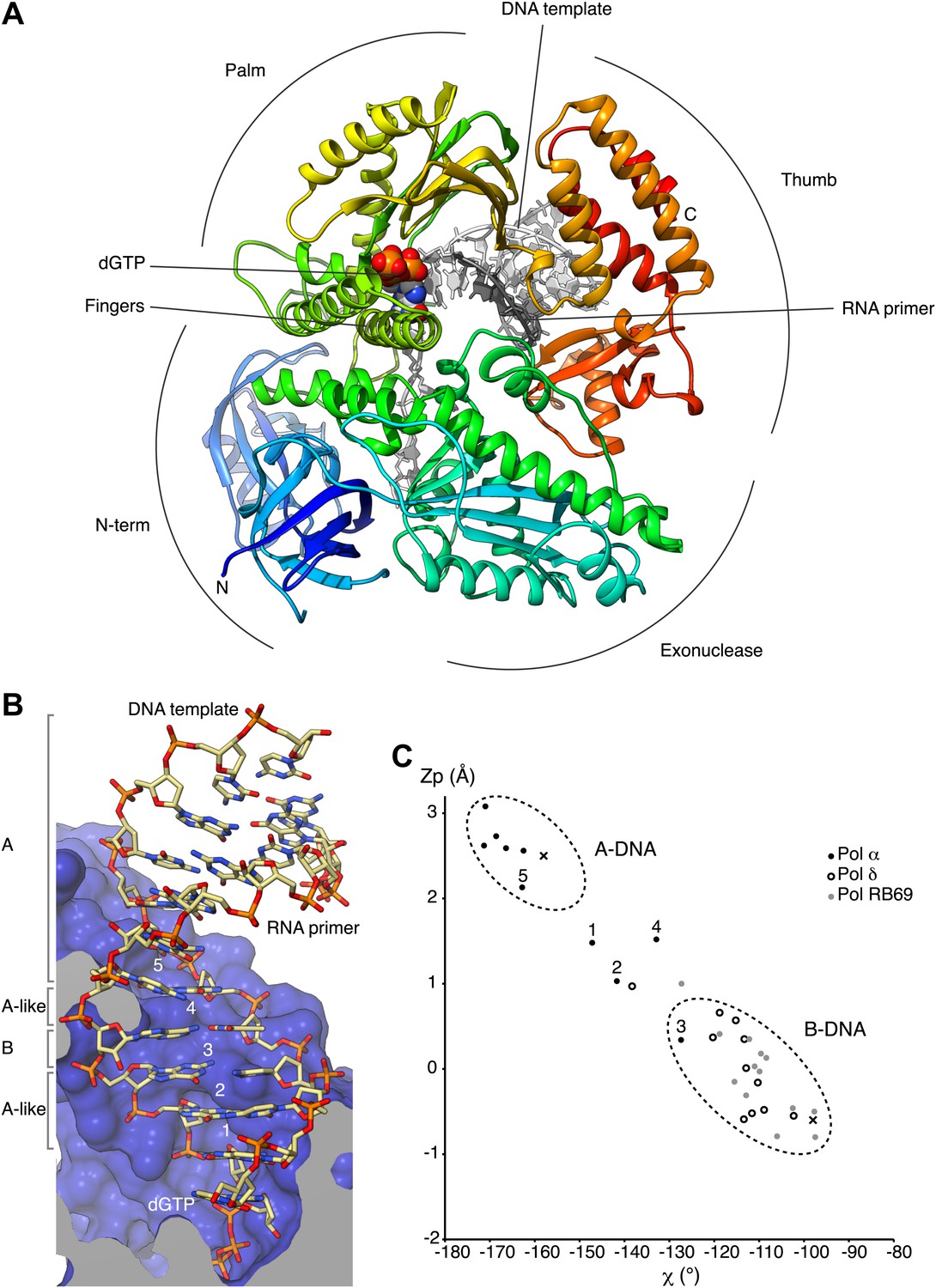
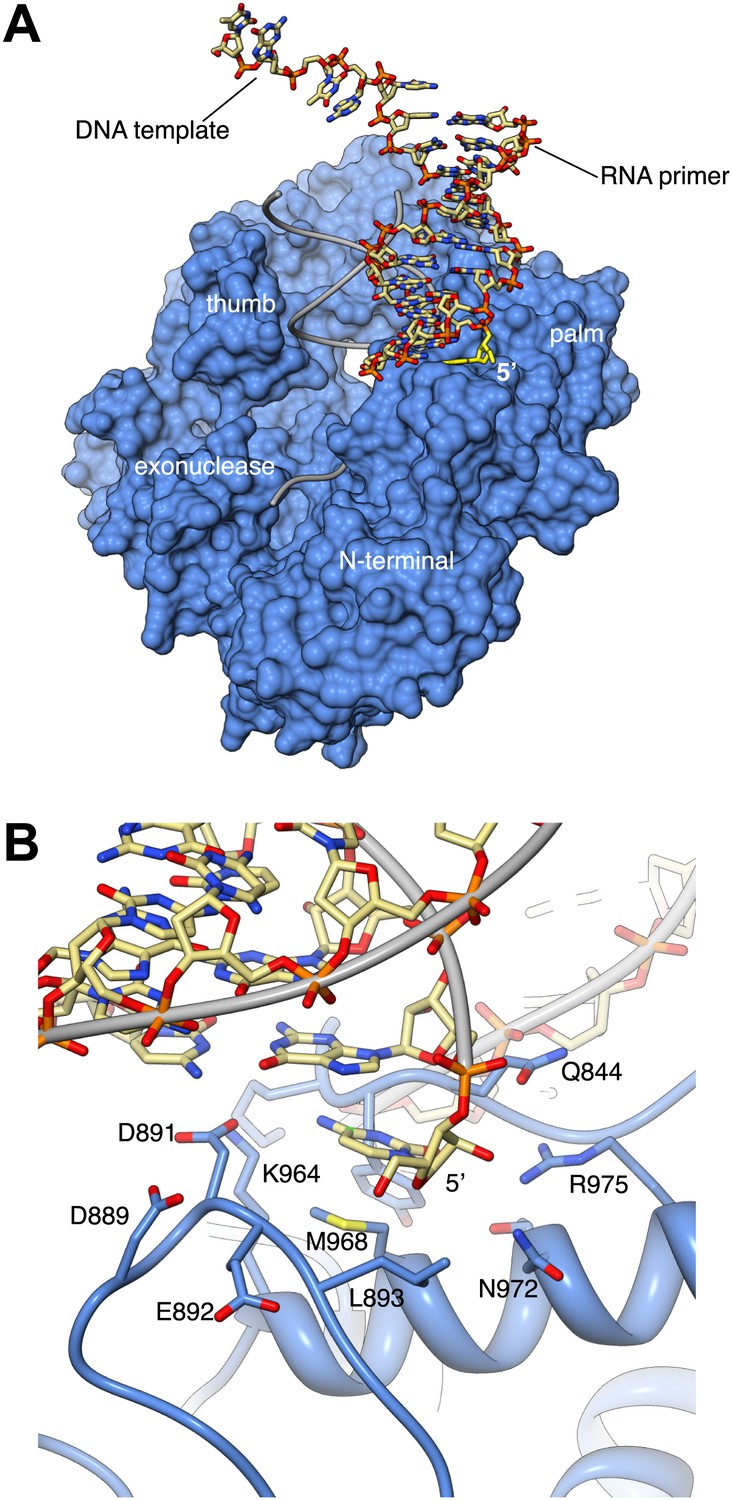
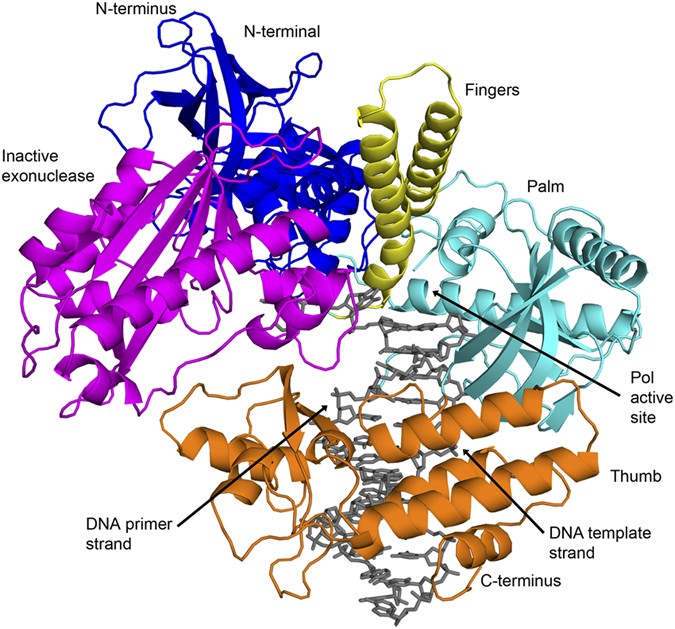
Activity and fidelity of human DNA polymerase α depend on primer structure - Journal of Biological Chemistry

The human CTF4-orthologue AND-1 interacts with DNA polymerase α/primase via its unique C-terminal HMG box | Open Biology

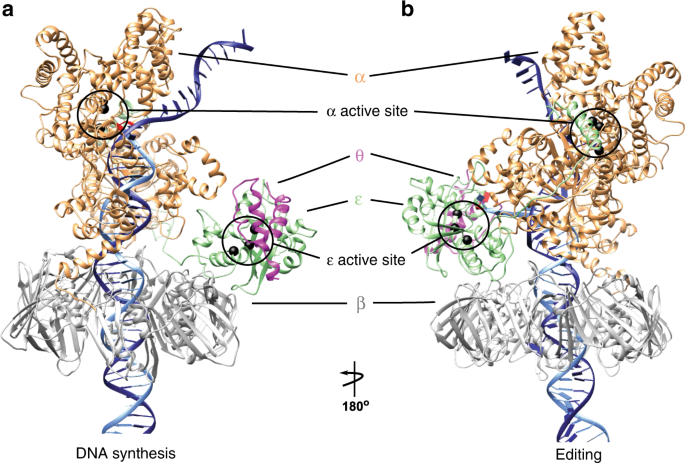
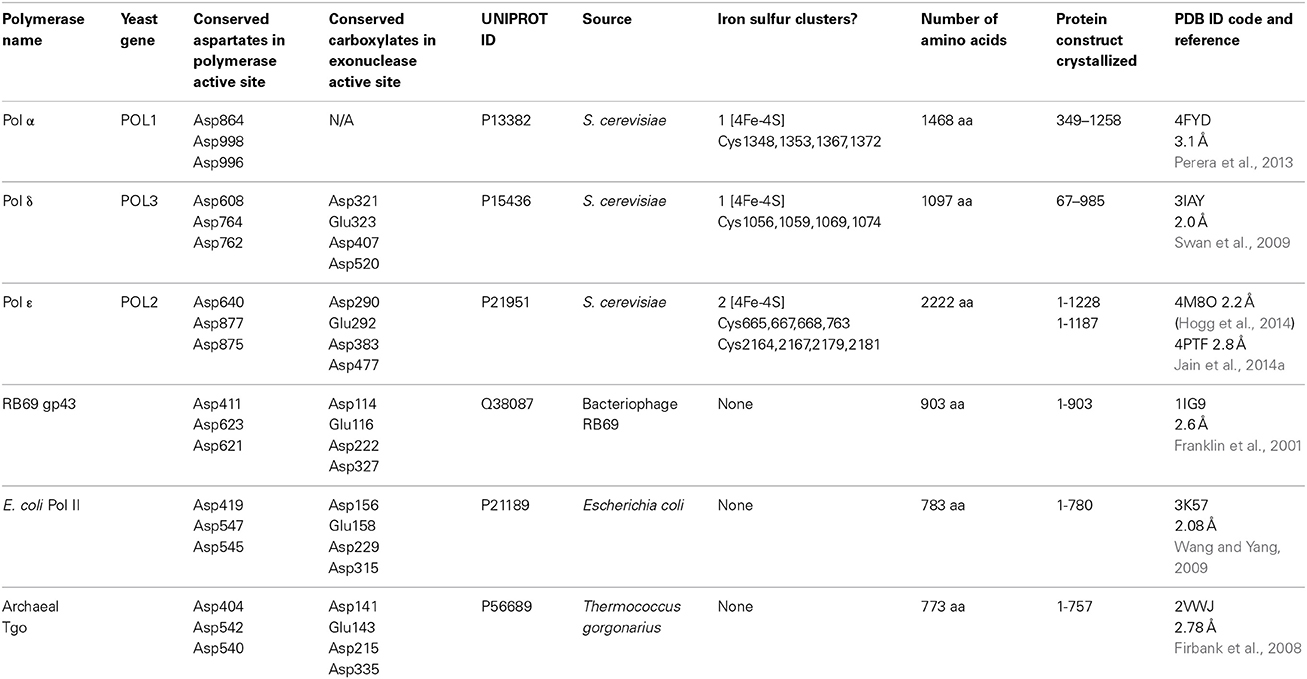
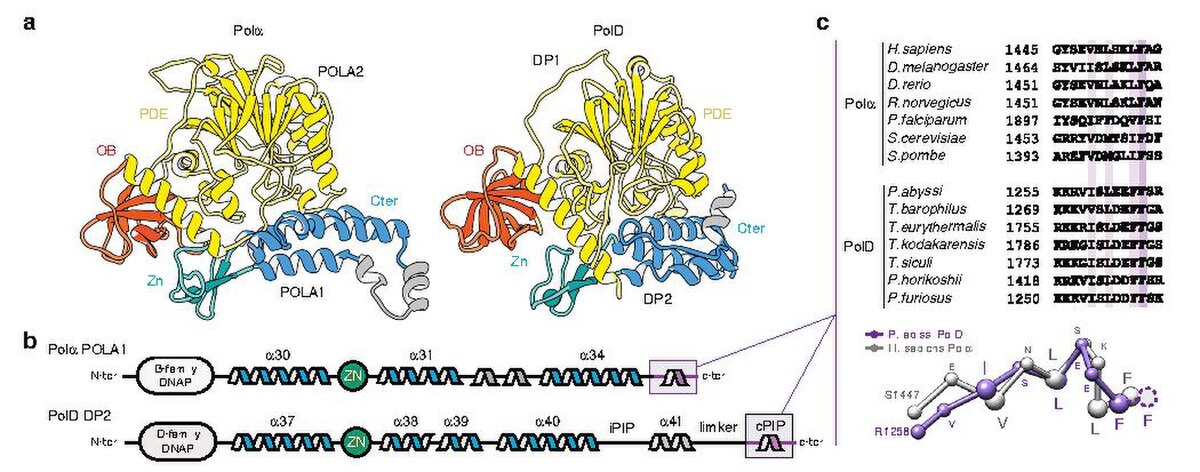
3D architecture of DNA Pol α reveals the functional core of multi‐subunit replicative polymerases | The EMBO Journal
Separable, Ctf4-mediated recruitment of DNA Polymerase α for initiation of DNA synthesis at replication origins and lagging-strand priming during replication elongation | PLOS Genetics
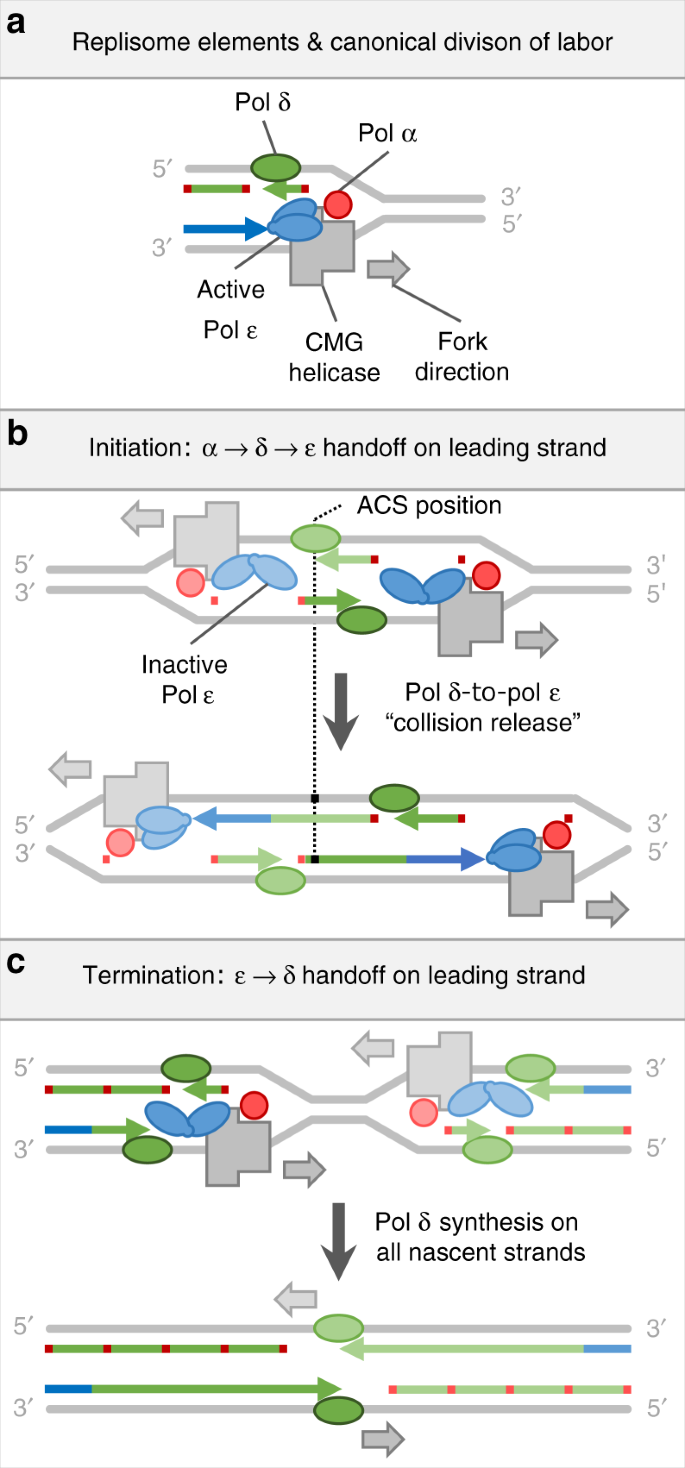
Roles for DNA polymerase δ in initiating and terminating leading strand DNA replication | Nature Communications

Replicative enzymes and ageing: importance of DNA polymerase alpha function to the events of cellular ageing - ScienceDirect

A Major Role of DNA Polymerase δ in Replication of Both the Leading and Lagging DNA Strands - ScienceDirect

3D architecture of DNA Pol α reveals the functional core of multi‐subunit replicative polymerases | The EMBO Journal

Structural basis of transcription: α-Amanitin–RNA polymerase II cocrystal at 2.8 Å resolution | PNAS